Delivering sustainable innovation for sterile packaging
A case study on Olympus adopting ฐีฒโฑนฑ๐ฐ์ยฎ with Renewable Attribution to incorporate certified renewable materials into packaging without impacting global manufacturing or the validation processes

Developing a sustainable healthcare packaging solution requires lowering environmental impact without compromising patient safety. This balance is challenging in a highly regulated, riskโaverse industry where every material change must be rigorously proven safe. Meaningful progress, therefore, depends on close collaboration across the value chain to ensure that sustainability does not come at the expense of safety.
Olympus Corporation, a global MedTech leader, has collaborated with บฃฝวด๓ษ๑ to introduce more environmentally responsible packaging for its single-use endotherapeutic devices worldwide. Starting in 2026, Olympus will incorporate บฃฝวด๓ษ๑โข ฐีฒโฑนฑ๐ฐ์ยฎ with Renewable Attribution into sterile packaging for over 100 device categories.1 This solution is linked to a certified mass balance approach to integrate renewable feedstocks that reduce reliance on fossil-derived raw materials while maintaining packaging performance. ฐีฒโฑนฑ๐ฐ์ยฎ with Renewable Attribution provides approximately a one-third reduction in CO2 emissions, delivering an immediate environmental gain. The first phase will launch at Olympus facilities in Japan and Vietnam, with plans to expand globally.
โThis collaboration with บฃฝวด๓ษ๑ directly supports Olympusโs Environment, Social and Governance (ESG) strategy of realizing a carbon-neutral society and circular economy. By introducing ฐีฒโฑนฑ๐ฐ์ยฎ with Renewable Attribution, we are accelerating our transition toward low-carbon and resource-efficient product design.โ
- Yuichi Morizane, Senior Director of the R&D Center of Sustainability Excellence at Olympus.
In this case study, we show how the collaboration between Olympus and บฃฝวด๓ษ๑ helped enable sustainable packaging design and material innovation, turning shared challenges into shared progress.
Challenge
Healthcare systems worldwide depend on plastics to deliver safe and effective care. Millions of tons of healthcare plastics are produced and consumed annually, and this number continues to grow.2 With this growth comes an inevitable increase in plastic waste and the emissions associated with its life cycle. Hospitals and healthcare providers are responding by introducing sustainability and green procurement criteria into public tenders, incentivizing suppliers to deliver packaging solutions that reduce environmental impact.3
Programs such as the Danish Nordic Criteria for More Sustainable Packaging for Healthcare Products4, the U.S. EPAโs Environmentally Preferable Purchasing Program5 and the NHS Net Zero Supplier Roadmap6 underscore the importance of packaging that supports circularity, recyclability and reduced carbon footprint. Industry leaders like Olympus are signaling the same shift by embedding sustainability into core business strategies and setting measurable targets for carbon neutrality and circular economy principles.7 For example, Olympus has committed to achieving net zero greenhouse gas emissions across its value chain by 2040 through collaboration with customers and suppliers.8
This alignment between procurement trends and industryโs ESG priorities suggests that sustainability is becoming a defining factor in competitive positioning.
At the same time, sterile packaging plays a critical role in patient safety, ensuring that medical devices remain free from contamination from the point of manufacture to the point of use. Sterile packaging must allow for effective sterilization and maintain sterility throughout the productโs life cycle. In practice, plastics remain the dominant choice for sterile packaging because their toughness and durability provide the most reliable protection. Because this performance is difficult to achieve with alternative materials, advancing the circularity of plastics themselves becomes essential for reducing environmental impact without compromising patient safety.
While integrating renewable or recycled raw materials into healthcare packaging solutions meaningfully contributes to a more circular economy, their incorporation into sterile packaging is limited due to the understandably stringent performance, quality and safety requirements of the sector. Preserving sterile barrier performance requires extensive, timeโconsuming and costly validation to prove its equivalence, efficacy and safety. These requirements present significant challenges to changing raw materials used in healthcare packaging. As a consequence, sterile medical device packaging solutions are predominantly made from virgin-grade, fossil-based raw materials without scalable, efficient and safe alternatives to transition to more circular raw materials.
Regulatory frameworks add another layer of complexity. In Europe, medical device packaging is regulated by the Medical Devices Regulation (MDR).9 Companies providing solutions to the European market must comply with standards that demonstrate compliance with the MDR. One key standard concerning healthcare packaging is ISO 11607, which stipulates that โThe source, history and traceability, especially recycled materials, shall be known and controlled to ensure that the preformed sterile barrier system and/or sterile barrier system will consistently meet the requirements of this document.โ10 Thus, for broad-based adoption by the value chain, it is crucial that innovations enhancing circularity are introduced into sterile packaging in a manner that seamlessly facilitates compliance with regulatory requirements, making this an extremely complex and challenging endeavor.
For these reasons, the healthcare packaging industry remains largely linear and dependent on virgin-grade, fossil-derived raw materials. Achieving a circular economy with scalable, safe and compliant solutions calls for systemic change and cross-industry collaboration.
Solution
The polymer industry faces growing pressure to reduce dependence on fossil resources while maintaining the performance and scale of existing products.
One practical and more cost-effective approach is to introduce renewable raw materials into established value chains rather than building entirely new systems.
While the technologies needed to make plastics from renewable feedstocks already exist, the global availability of renewable feedstocks remains insufficient to support dedicated, largescale infrastructure for entirely renewable polymer production. According to PlasticsEurope11, bio-based and bio-attributed plastics represented only about 1% of Europeโs total plastics production in 2024. Concurrently, typically less than 5% of the input can be substituted due to cost of alternative raw materials and technology limitations12. Although alternative feedstocks could be high volumes in absolute terms, claiming products with less than 5% bio-based content would not be attractive enough to drive demand.
Therefore, to accelerate the integration of renewable raw materials without waiting for fully segregated supply chains, the polymer industry employs the mass balance approach.13 Under the mass balance approach, renewable and fossil-based feedstocks can be co-processed in existing manufacturing facilities, and the renewable quantities can be attributed to products at meaningful levels (for example, 30% attribution). Rather than physical segregation, the attribution of renewable raw materials is managed through a certified and audited accounting system.
Learn how ฐีฒโฑนฑ๐ฐ์ยฎ with Renewable Attribution can help make it possible at
tyvek.com/RenewableAttribution
Transitioning to more circular raw material use
Polymer companies have introduced renewable raw materials, such as first- and second-generation feedstocks, ranging from edible agricultural products (e.g., sugarcane) to waste and residues from the food industry (e.g., vegetable oils). While there are many technologies for incorporating these renewable feedstocks to produce polyolefins, the most widely adopted method is a bio-naphtha steam cracking pathway. 14 15
Used cooking oil (UCO) stand out as a promising resource among renewable feedstock alternatives because of its availability and compability with existing production infrastructure. Similar to fossil feedstock, UCO can be introduced into the polyolefin production process after comprehensive pre-treatment (removal of impurities, such as water, trace metals and other residues) and refining to meet stringent feedstock specifications. The refined UCO then undergoes hydrodeoxygenation, a process where oxygen is removed and the oil is transformed into a mixture of saturated linear and branched-chain hydrocarbons, known as bio-naphtha. This is a high-quality feedstock that can be processed in steam crackers in an identical manner to fossil-derived naphtha. 16 17 18
Steam cracking is a high-temperature process that breaks down hydrocarbon feedstocks, such as fossil and bio-naphtha, into smaller molecules, primarily ethylene and propylene. Ethylene is the simples olefin, composed of a two-carbon backbone with a carbon-carbon double bond and four attached hydrogen atoms. It is a colorless, flammable gas at normal temperature and pressure, and its double bond makes it highly reactive and widely used to produce plastics like polyethylene. Because ethylene is ethylene, whether derived from fossil or renewable raw materials, the use of renewable feedstocks represents a compelling approach to help enable sustainability in healthcare packaging. In addition, since etyhlene cannot be distinguished by source (i.e., fossil or renewable-derived), a mass balance approach is needed to attribute the quantities of renewable-derived ethylene to final products on a bookkeeping basis. 19
บฃฝวด๓ษ๑ sources high-density polyethylene (HDPE) that meets stringent specifications.
Renewable raw materials are attributed to HDPE using the mass balance approach through Sustainability Declarations under the ISCC PLUS system, a fully traceable system, ascertaining responsible accounting practices.
Renewable raw materials are attributed to HDPE using the mass balance approach through Sustainability Declarations under the ISCC PLUS system, a fully traceable system, ascertaining responsible accounting practices.
Tracking renewable raw materials from feedstock through packaging
A traceable path through a mass balance approach.
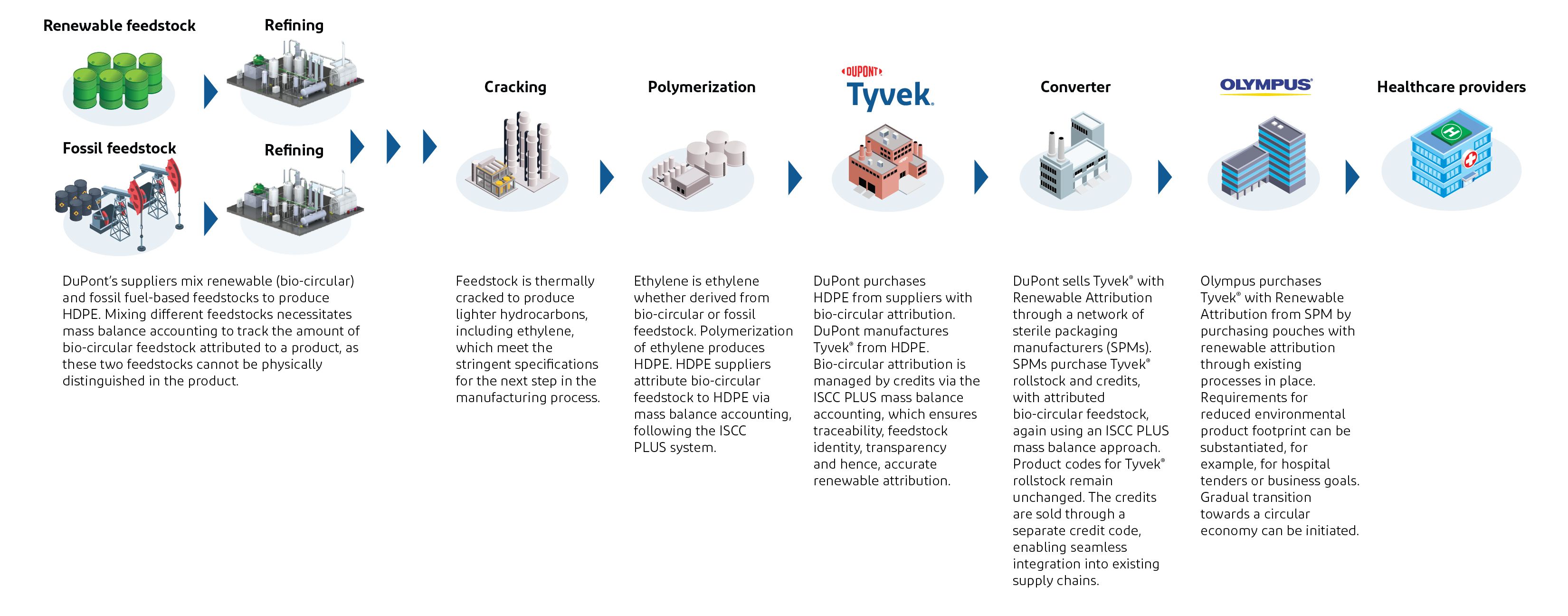
บฃฝวด๓ษ๑ manufactures ฐีฒโฑนฑ๐ฐ์ยฎ rollstock using existing processes and passes on Sustainability Declarations further downstream in the value chain. In order to maintain clarity and transparency, these transactions of product and Sustainability Declarations are enabled through a new product extension called ฐีฒโฑนฑ๐ฐ์ยฎ with Renewable Attribution, launched in November 2024.
ฐีฒโฑนฑ๐ฐ์ยฎ with Renewable Attribution is sold through sterile packaging manufacturers (SPMs) that purchase the rollstock material together with the corresponding Sustainability Declaration. Medical device manufacturers (MDMs), like Olympus, purchase ฐีฒโฑนฑ๐ฐ์ยฎ with Renewable Attribution through existing channels, along with Sustainability Declarations that demonstrate the attribution of renewable share.
Thus, this approach introduces renewable raw materials into the current supply chain without changing ฐีฒโฑนฑ๐ฐ์ยฎ โs material specifications or manufacturing processes, and can be transferred (sold) downstream provided the entire chain is ISCC PLUS certified. Sustainability Declarations, audits and documentation maintain transparency and control, providing accurate renewable attribution from feedstock origin up to the final packaging.
Impact
Olympus has taken a significant step toward addressing sustainability challenges in sterile healthcare packaging by incorporating ฐีฒโฑนฑ๐ฐ์ยฎ with Renewable Attribution into its single-use device portfolio. ฐีฒโฑนฑ๐ฐ์ยฎ is the first permeable sterile packaging solution linked to certified renewable raw materials, delivering a meaningful reduction in carbon footprint while maintaining the performance and reliability required in healthcare. Designed as a drop-in solution, ฐีฒโฑนฑ๐ฐ์ยฎ with Renewable Attribution provides immediate, seamless integration into the global supply chains of Olympus products already using ฐีฒโฑนฑ๐ฐ์ยฎ , without requiring changes to specifications or regulatory documentation.
In the short term, Olympus is already gaining benefits. The adoption of ฐีฒโฑนฑ๐ฐ์ยฎ with Renewable Attribution was achieved rapidly because Olympus determined that no extensive revalidation data sets were required.
Compared with the typical adoption time for a new sterile packaging material, time to market was significantly reduced, allowing Olympus to introduce a more sustainable solution much sooner. The fact that ฐีฒโฑนฑ๐ฐ์ยฎ with Renewable Attribution is an ISCC PLUS certified mass balance solution, Olympus determined that the need for typical extensive physical material tests could be eliminated because the physical properties remain unchanged. Olympus is also realizing immediate environmental gains with the shift to ฐีฒโฑนฑ๐ฐ์ยฎ with Renewable Attribution, which is expected to reduce the materialโs carbon footprint by roughly one third, providing a significant contribution toward Olympusโs climate objectives.
In the long term, Olympusโs continued integration of ฐีฒโฑนฑ๐ฐ์ยฎ with Renewable Attribution supports its broader sustainability strategy, particularly its commitment to achieving a โcarbon neutral society and circular economy.โ20 By choosing ฐีฒโฑนฑ๐ฐ์ยฎ with certified renewable feedstocks via a mass balance approach, Olympus is advancing its long-term carbon neutral goals while maintaining the same high standards of packaging performance and patient safety that the industry requires.
Hospitals and healthcare systems are increasingly expecting more circular, resource-efficient solutions.
โบฃฝวด๓ษ๑ are seeing increasing emphasis on sustainability in U.S. and European tenders. This is also reflected by a more than two-fold increase in ESG-related customer inquiries over the past few years. Solutions like ฐีฒโฑนฑ๐ฐ์ยฎ with Renewable Attribution are helping us address emerging expectations for recycled content and bio-based materials in primary sterile packaging, something that wasnโt feasible with sterile packaging until now.โย
- Yuichi Morizane, Senior Director of the R&D Center of Sustainability Excellence at Olympus.
This shift signals both an immediate advantage in meeting current procurement expectations and a long-term competitive benefit as sustainability criteria continue to strengthen globally. This case study shows how Olympus leveraged ฐีฒโฑนฑ๐ฐ์ยฎ with Renewable Attribution to achieve meaningful carbon reductions quickly and efficiently, setting a benchmark for sustainable innovation in healthcare packaging.
References
1 Olympus Corporation, Olympus Commits to Advancing Sustainable Packaging Enabled by บฃฝวด๓ษ๑โข ฐีฒโฑนฑ๐ฐ์ยฎ with Renewable Attribution (available at: ). Olympus website, 2025. Accessed April 3, 2026.
2 Healthcare Plastics Recycling Council (HPRC), WHY HPRC: Inspiring and Enabling Plastic Recycling Solutions in Healthcare (available at: ). HPRC website, 2026. Accessed April 3, 2026.
3 MedTech Europe, Decarbonising Healthcare: How a Competitive Medical Technology Industry Can Contribute (available at: ). MedTech Europe website, 2025. Accessed April 3, 2026.
4 Danske Regioner, Nordic Criteria for More Sustainable Packaging (available at: ). Danske Regioner publication, 2023. Accessed April 3, 2026.
5 U.S. Environmental Protection Agency (EPA), About the Environmentally Preferable Purchasing Program (available at: ). EPA website, 2025. Accessed April 3, 2026.
6 NHS England, NHS Net Zero Supplier Roadmap (available at: ). NHS publication, 2023. Accessed April 3, 2026.
7 Olympus Corporation, What Stakeholders Are Doing to Achieve Healthcare Sustainability (available at: ). Olympus white paper, 2024. Accessed April 3, 2026.
8 Olympus Corporation, Focus Area 5: Carbon Neutral Society and Circular Economy (available at: ). Olympus website, 2026. Accessed April 3, 2026.
9 European Union, Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017 on Medical Devices (available at: ). EUR-Lex, 2026. Accessed April 3, 2026.
10 International Organization for Standardization (ISO), Packaging for Terminally Sterilized Medical Devices (ISO 11607) (available at: ). ISO standard, 2019. Accessed April 3, 2026.
11 PlasticsEurope, Plastics โ The Fast Facts 2025 (available at: ). PlasticsEurope publication, 2025. Accessed April 3, 2026.
12 Renewable Carbon Publications, Importance of Mass Balance and Attribution (MBA) for the Conversion of the Chemical Sector to Alternative Carbon Sources (available at: ). Renewable Carbon Publications, 2025.
13 International Sustainability and Carbon Certification (ISCC), Accelerating the Transition to a Circular Economy (available at: ). ISCC website, 2026.
14 Renewable Carbon Publications, Alternative Naphtha โ Technologies and Market Status and Outlook (available at: ). Renewable Carbon Publications, 2024. Accessed April 3, 2026.
15 S&P Global, Bio Chemicals 2025 โ An Evolving Sustainability Stream (available at: ). S&P Global report, 2025. Accessed April 3, 2026.
16 ScienceDirect, Biomass to Olefins: Cracking of Renewable Naphtha (available at: ). Chemical Engineering Journal, 2011. Accessed April 3, 2026.
17 Kumar, A., et al., Valorization of Used Cooking Oil: Challenges, Current Developments, Life Cycle Assessment and Future Prospects (). Discover Sustainability, 2025. 6(1): p. 119. Accessed April 3, 2026.
18 Neste, The ABC of UCOs โ What Are Used Cooking Oils and How Can They Be Used in a More Circular Economy? (available at: ). Neste website, 2023. Accessed April 3, 2026.
19 International Sustainability and Carbon Certification (ISCC), Mass Balance Explained (available at: ). ISCC website, 2024. Accessed April 3, 2026.
20 Olympus Corporation, Focus Area 5: Carbon Neutral Society and Circular Economy (available at: ). Olympus website, 2026. Accessed April 3, 2026.
บฃฝวด๓ษ๑ believes this information to be reliable. It may be subject to change as additional knowledge and experience are gained. It is not intended as a substitute for any testing you may conduct to determine for yourself the suitability of our products and information for your particular purpose. Since conditions for use are outside the บฃฝวด๓ษ๑โs control, DUPONT DE NEMOURS, INC. AND ITS AFFILIATES MAKE NO WARRANTIES, EXPRESSED OR IMPLIED, INCLUDING BUT NOT LIMITED TO WARRANTIES OF MERCHANTABILITY OR FITNESS FOR A PARTICULAR PURPOSE AND ASSUMES NO LIABILITY IN CONNECTION WITH ANY USE OF THIS PRODUCT AND INFORMATION. This information is not intended as a license to operate under or a recommendation to infringe any trademark, patent or technical information of บฃฝวด๓ษ๑ or other persons covering any material or its use.
ยฉ2026 บฃฝวด๓ษ๑. All rights reserved. บฃฝวด๓ษ๑โข, the บฃฝวด๓ษ๑ Oval Logo, and all trademarks and service marks denoted with โข, SM or ยฎ are owned by affiliates of บฃฝวด๓ษ๑ de Nemours, Inc. unless otherwise noted.